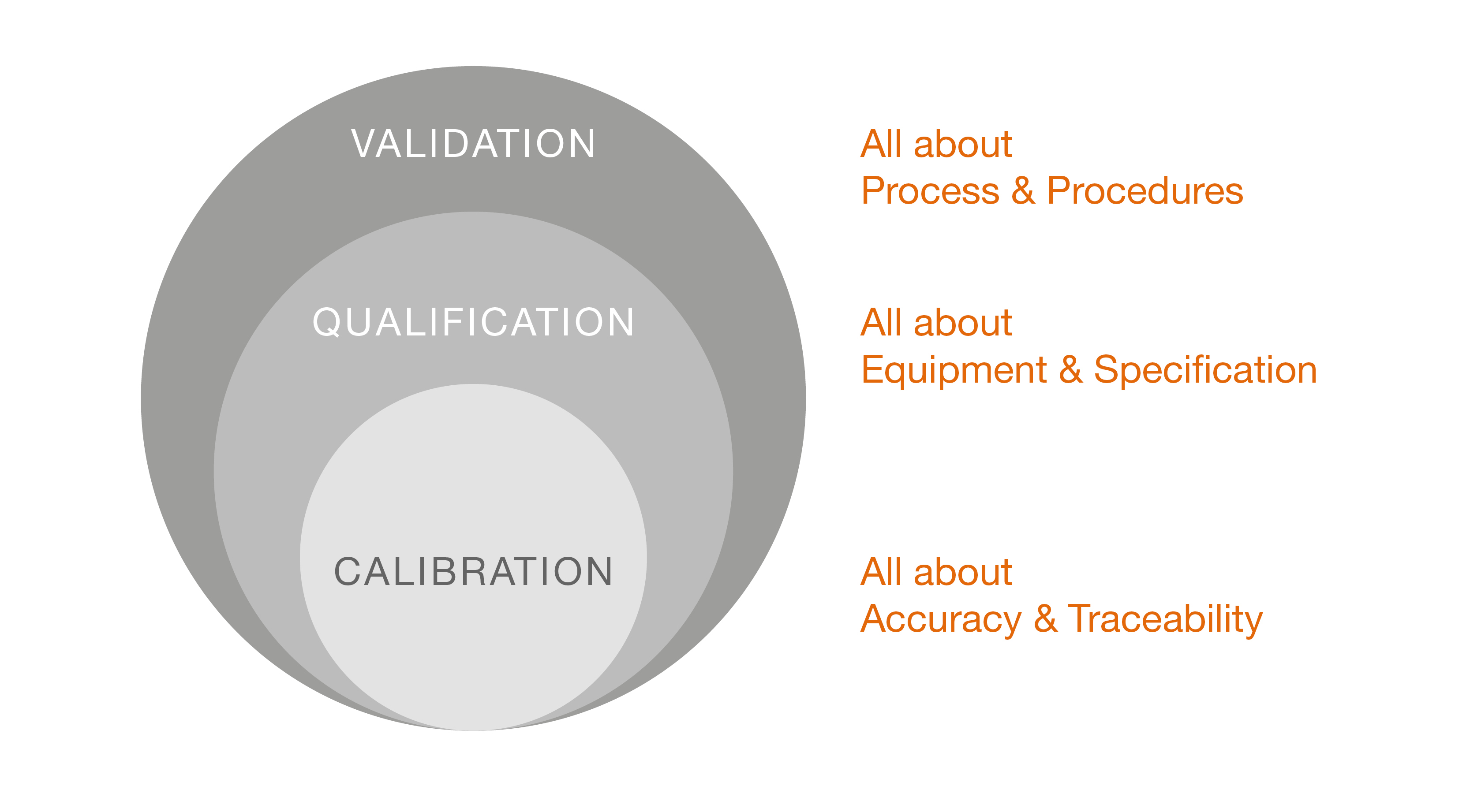
Validation is the documented process of verifying that a system, process, equipment, or product reliably meets the specific requirements and quality standards. This process ensures that all critical parameters are controlled and consistently produce reproducible results to guarantee both the safety and effectiveness of the final product. Thus, validation is the documented evidence that all procedures, processes, facilities, systems, materials, and equipment comply with GxP requirements and deliver the expected results.